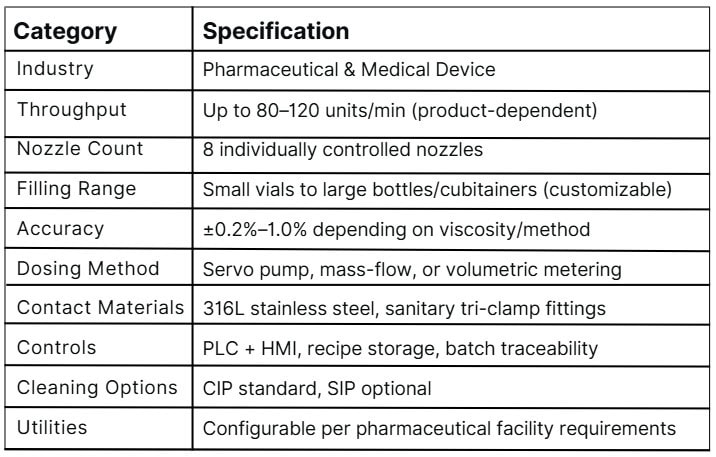
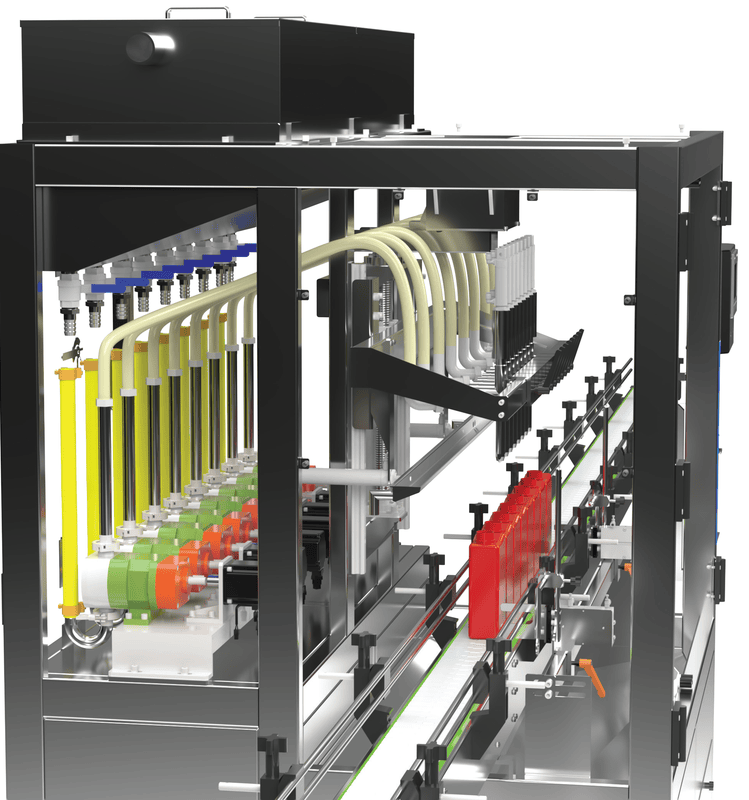
The Machinelab 8-Nozzle Servo-Driven Filling Machine is engineered for the rigorous requirements of pharmaceutical and medical device liquid processing. Designed for hygienic, accurate, and repeatable filling across a wide range of container sizes, it delivers reliable performance in fully regulated environments.
Built in Ireland with servo-driven dosing, GMP-compliant construction, and sanitary product-contact components, it is ideally suited for sterile solutions, diagnostic reagents, buffer liquids, irrigation fluids, nutraceutical syrups, and other medical-grade formulations.
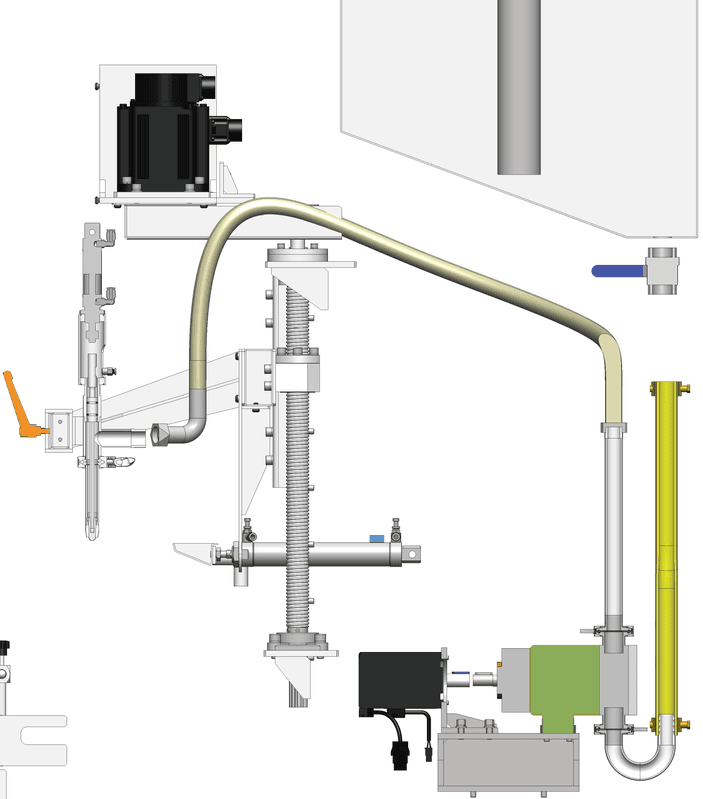
Delivers fast, hygienic, and repeatable filling with independent servo-driven nozzles designed for sterile and low-to-medium viscosity pharmaceutical liquids.
Built with 316L stainless steel product-contact surfaces and hygienic flow paths, fully suited for Grade C/D cleanroom operation.
Advanced servo flow profiling ensures stable, turbulence-free filling with precise volume control across all nozzles.
Supports validated Clean-in-Place with optional Steam-in-Place sterilization for GMP, FDA, and EU Annex 1 environments.
Siemens/Allen-Bradley PLC with touchscreen HMI enables full recipe control, batch parameters, and seamless SCADA/MES integration.
Compact stainless-steel enclosure engineered for cleanroom efficiency with easy maintenance access.